Traceability failure compromises patient safety faster than almost any other procedural error in the Central Sterile Services Department. While sterilisation equipment performs the heavy lifting of pathogen elimination, the label carries the evidentiary burden because it proves the process occurred. A sterile pack without precise data is clinically indistinguishable from a contaminated one. This reality places immense pressure on CSSD managers and theatre staff to maintain flawless records while managing high volume throughput.
Infection control relies on the certainty that every instrument used in a procedure is sterile. The Australian Commission on Safety and Quality in Health Care reports that healthcare associated infections (HAIs) account for approximately two million lost bed days annually in Australia. Effective labelling serves as the primary defence against this statistic. It transforms a plastic pouch into a traceable medical asset.
The regulatory baseline for Australian facilities
Compliance in Australia hinges on strict adherence to AS/NZS 4187:2014. This standard mandates rigorous traceability for all Reprocessable Medical Devices (RMDs). It requires health service organisations to track an instrument from the patient procedure back to the specific steriliser and cycle that processed it.
Many facilities struggle with the practical application of this standard because the volume of instruments is overwhelming. Staff must record the steriliser identification number, the cycle number, and the date of processing for every single item. This data chain allows infection control teams to recall specific batches if a biological indicator fails or if a steriliser malfunctions.
Essential data points for clinical safety
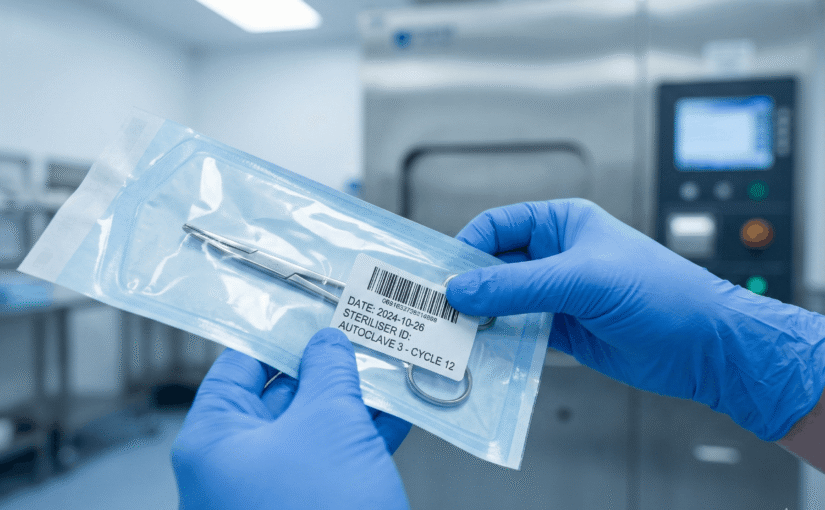
A compliant label must convey specific information to ensure seamless tracking. Omission of even one data point breaks the chain of custody and renders the pack non compliant during an audit.
The label must clearly display the steriliser identification code. This allows staff to pinpoint exactly which machine processed the equipment. Following this, the cycle number must appear to identify the specific load. This precision is vital because a machine may pass a morning leak rate test but fail a subsequent vacuum test.
The processing date dictates the shelf life calculation and stock rotation. Staff should also include the packer identification. This fosters accountability and allows managers to identify training gaps if packs consistently fail visual inspection due to poor wrapping techniques.
Strategic placement preserves barrier integrity
Where a staff member places a label matters as much as what the label says. The sterile barrier system is fragile.
Staff must apply labels strictly to the plastic laminate side of the pouch. The paper side of a sterilisation pouch functions as a breathing membrane which allows the sterilising agent to enter and exit. Adhesives applied to this paper surface can clog the pores and prevent the sterilising agent from penetrating the pack.
Applying a label to the paper also creates a risk of tearing during opening. If the paper tears, fibres can contaminate the instruments inside. Placing the label on the plastic film ensures the data remains visible without compromising the structural or functional integrity of the packaging material.
Material science and chemical safety
Standard office stationery has no place in a sterile field. Labels used in CSSD environments must withstand extreme conditions without degrading.
Steam sterilisation involves temperatures up to 134°C and significant moisture changes. Hydrogen peroxide plasma and ethylene oxide cycles introduce chemical variables that interact with standard adhesives. Mermed engineers labels specifically to resist these stressors.
Chemical migration poses a silent risk to patients. Inks used on sterilisation labels must be non toxic and stable. If an ink runs or off gases during the heating process, it can penetrate the pouch and contaminate the surgical instruments. Facilities must verify that their labelling consumables are validated for the specific sterilisation modality they use.
Improving workflow efficiency
Manual data entry slows down the decontamination cycle and invites human error. Writing details by hand on every pouch is inefficient and creates legibility issues that complicate records.
labelling guns and automated printing systems reduce the cognitive load on staff. These tools ensure that every label is legible and contains consistent data fields. This consistency speeds up the checking process in the operating theatre. A nurse can verify the sterility status of an instrument in seconds rather than deciphering handwriting.
Colour coding offers another layer of efficiency. Using specific label colours for different departments or urgency levels allows staff to visually sort packs quickly. This visual management system reduces retrieval time when a specific instrument is needed urgently in theatre.
Handling expiry and stock rotation
Sterility maintenance is event related rather than time related, but expiration dates remain a critical component of inventory management.
Labels must clearly indicate the expiry date based on the facility policy. This supports the “First In, First Out” principle of stock rotation. Staff can easily identify which packs need to be used first, which reduces wastage from expired stock.
When a pack reaches its expiration date, the facility must reprocess it. Clear labelling prevents the accidental use of expired equipment. This protects the patient from potential infection and protects the facility from liability.
Sterile labelling solutions provide precision and safety
Effective sterilisation labelling is not merely an administrative task. It represents the final check in a complex safety system.
By strictly adhering to AS/NZS 4187 guidelines and using purpose-built labelling solutions, facilities protect their patients and their practitioners. Precision in the CSSD translates directly to safety in the operating theatre.