A single unrecorded sterilisation cycle compromises an entire week of patient care. Infection control breaches remain the silent threat in dental operatories, capable of closing practices and ending careers.
The Australian Health Practitioner Regulation Agency (AHPRA) reported 1,446 notifications regarding dental practitioners in the 2022/23 financial year, and infection control complaints continue to feature prominently in regulatory actions. Documentation serves as the only defence against these allegations.
Dental professionals operate in high pressure environments where cognitive load peaks during complex procedures. Clinical staff juggle patient anxiety, technical precision, and strict time constraints. In this context, a label acts as more than a sticker. A label functions as a critical control point. Precision in labelling transforms abstract safety concepts into tangible, auditable evidence. This approach aligns dental clinic best practices with the rigorous standards seen in hospital operating theatres.
Why do dental clinic best practices hinge on traceability?
Regulatory bodies demand evidence that instruments used on a patient were sterile at the time of use. If your clinic is still using AS/NZS 4815:2006, you’re working under an outdated framework. AS 5369:2023 now serves as the modernised, risk-based operational standard for infection control and sterilisation in Australia. Unlike previous standards, AS 5369 applies broadly to all healthcare environments, including dental clinics.
AS 5369 mandates detailed records of each sterilisation cycle, including load identification, operator initials, and sterilisation cycle data. You cannot simply trust the autoclave passed. You must prove it passed.
Manual tracking often fails because human error is inevitable during busy turnover periods. A robust labelling system bridges this gap. When staff apply a label containing the cycle number, steriliser number, and date to every pouch, they create an unbroken chain of custody. This data must link back to the patient record. If a patient alleges a cross-infection occurring six months post-treatment, the practice can retrieve the specific cycle data that cleared the instruments used in that appointment.
Traceability protects the practitioner as much as the patient. The Dental Board of Australia expects strict adherence to these protocols. Failure to produce records suggests failure to sterilise. Mermed produces labelling solutions that integrate these variables into the daily workflow without adding time to the process.
How does visual logic prevent medication errors?
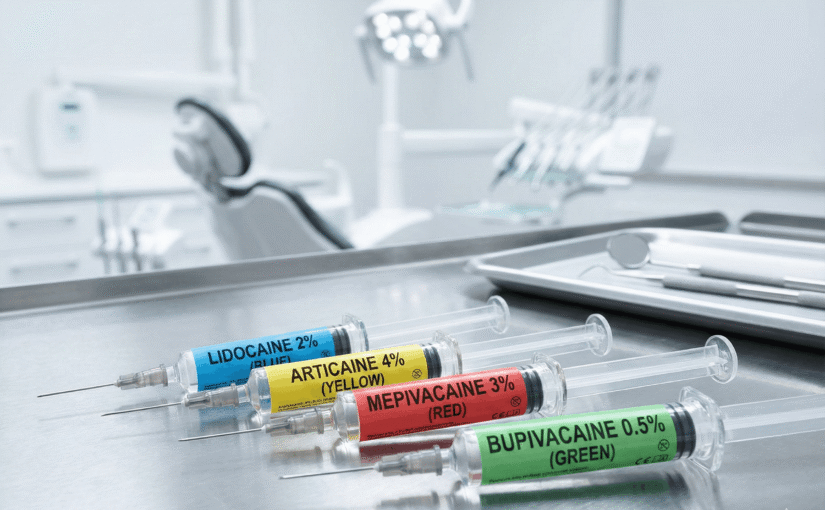
Dental operatories contain clear liquids that look identical but act differently. Sodium hypochlorite, local anaesthetic, and saline solution often sit in proximity on the bracket table. A mix-up here leads to catastrophic soft tissue trauma or systemic toxicity. The Australian Commission on Safety and Quality in Health Care (ACSQHC) identifies medication safety as a national priority, yet dental practices often rely on memory or temporary markers.
Standardised labelling removes the guesswork. Pre-printed labels for specific dental fluids utilise colour coding and clear text to signal contents immediately. This visual logic bypasses the need for the clinician to analyse a syringe mid-procedure. The brain recognises the colour pattern faster than it reads text.
Implementing a “labelling at the source” protocol ensures that any medicine drawn up is identified immediately. This reduces the cognitive burden on the dentist and the dental assistant. They can focus on the procedure because the safety infrastructure supports their decision making.
What is the financial risk of poor compliance?
An audit failure costs more than the fine. It costs the practice its reputation and operational days. When a state health department investigates a breach, the practice may face closure until they can demonstrate compliance. Revenue stops while overheads continue.
Investing in high quality labelling materials acts as an insurance policy for business continuity. Generic office labels often fail under the extreme conditions of steam sterilisation. If a label detaches inside the autoclave chamber, it can damage the equipment, leading to expensive repair bills and downtime. If the ink runs or fades, the legal record becomes void.
Mermed designs labels specifically for the Australian healthcare environment. These materials withstand the heat, moisture, and pressure of sterilisation cycles. They adhere to wraps and pouches without compromising the barrier system. Choosing purpose engineered materials demonstrates a commitment to quality assurance that auditors respect.
How can hospital grade solutions fit the dental workflow?
Many dental practice owners believe hospital grade traceability is too complex for a small clinic. This assumption leads to dangerous workarounds. However, AS 5369 is now closely aligned with hospital standard AS/NZS 4187, ensuring consistency between hospital sterilisation practices and primary care facilities. The goal is to adapt the rigour of the Central Sterile Services Department (CSSD) to the footprint of a sterilisation room.
Mermed partners with dental clinics to implement systems that fit their specific volume. Because every clinic layout differs, the solution must be flexible. Standardised documentation books, scanning guns, and double adhesive labels streamline the process. Staff simply peel the label from the pouch and affix it to the patient file. This action takes seconds but secures the compliance loop permanently.
Adopting these systems signals to patients that their safety drives every decision. While patients may not understand the mechanics of an autoclave, they notice the professionalism of a barcoded, tracked sterile pack. It builds trust. It reinforces the value of the care provided.
Shift from assumption to verification
Precision involves more than the drill. True clinical precision encompasses the entire ecosystem of care, from the sterilisation room to the patient record. Labelling provides the structural integrity for this ecosystem. It ensures that every instrument, medication, and file communicates truth.
Dental clinic best practices require a shift from assumption to verification. By transitioning to the AS 5369 standard and implementing rigorous labelling protocols, practices reduce the risk of adverse events and regulatory penalties while driving operational excellence. Mermed supports this transition with Australian engineered solutions designed for the realities of clinical practice. Safety is not a variable. It is the foundation.